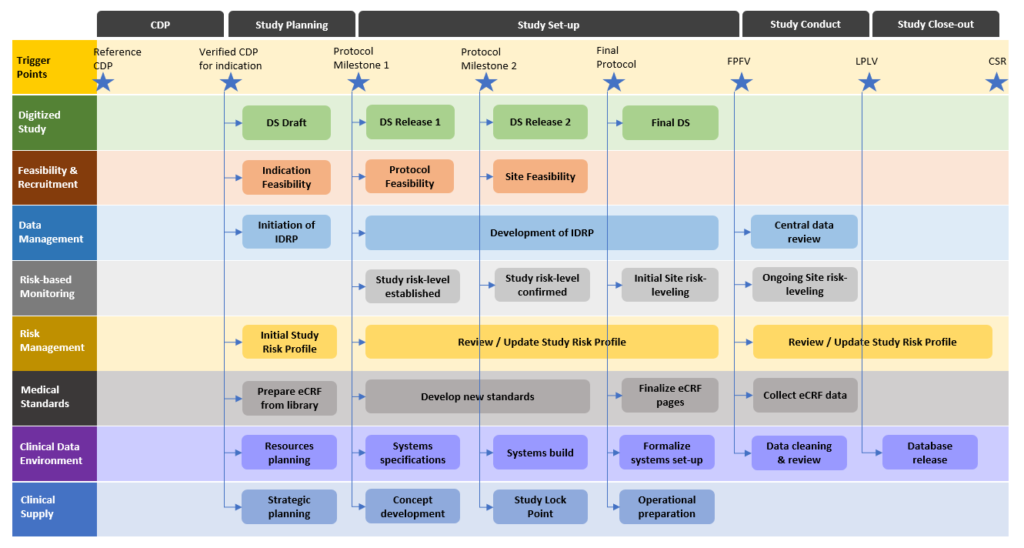
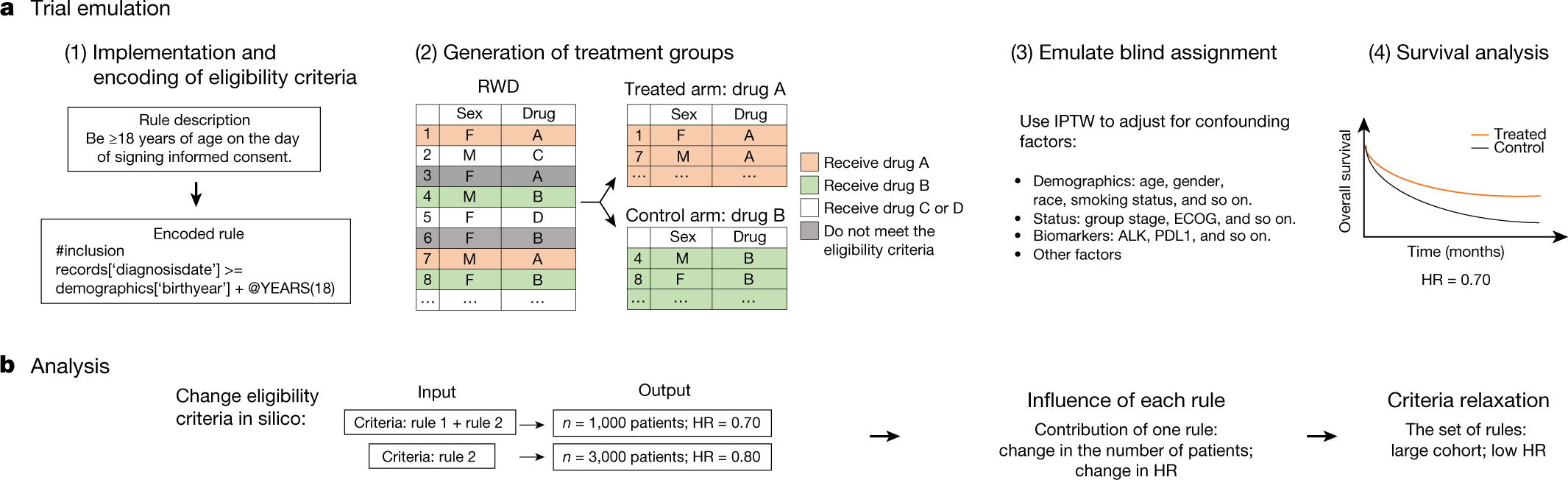
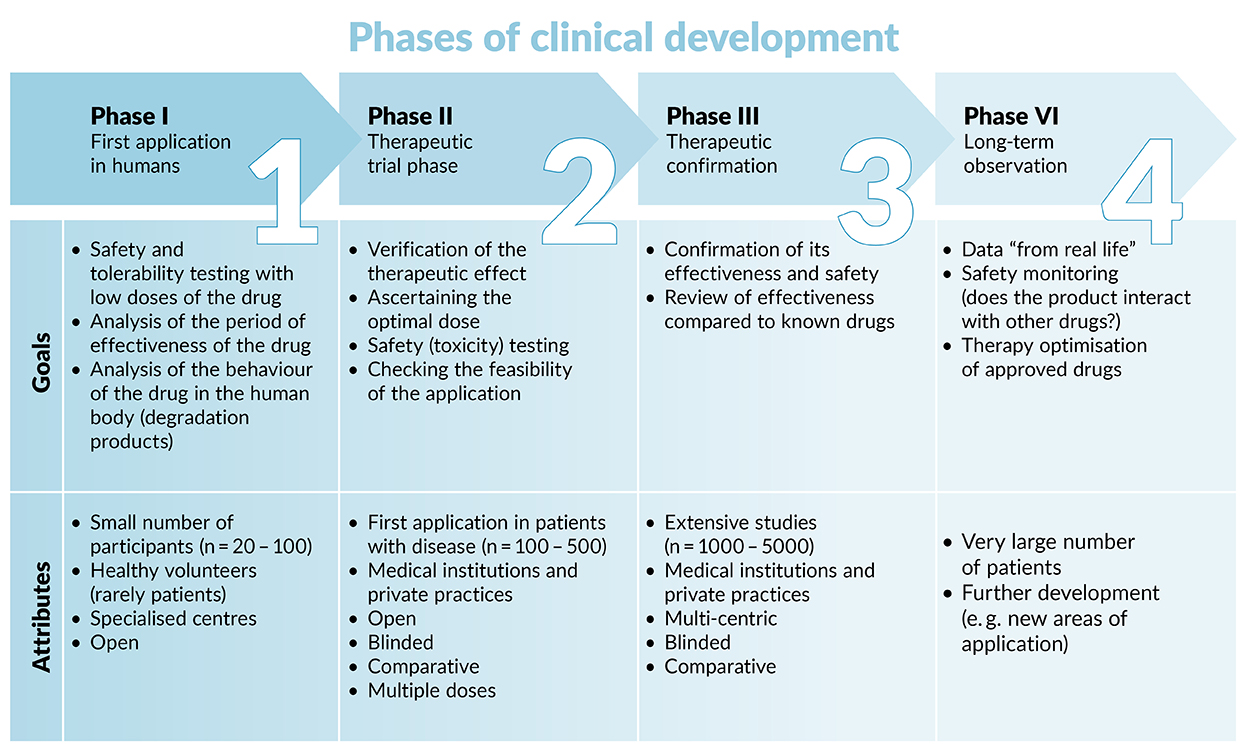
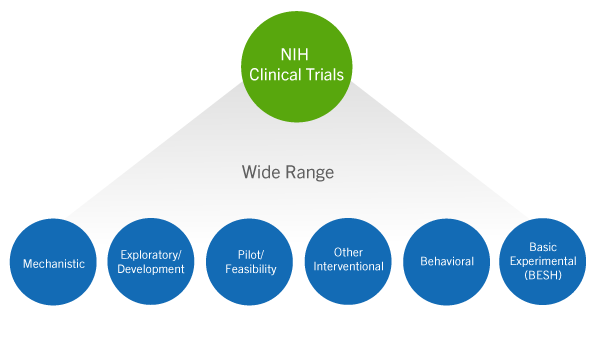
Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension - The Lancet Digital Health

Recruitment of Black Adults into Cardiovascular Disease Trials | Journal of the American Heart Association

Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension - The Lancet Digital Health

Assessing the detection, reporting and investigation of adverse events in clinical trial protocols implemented in Cameroon: a documentary review of clinical trial protocols – topic of research paper in Clinical medicine. Download

Early phase clinical trials extension to guidelines for the content of statistical analysis plans | The BMJ
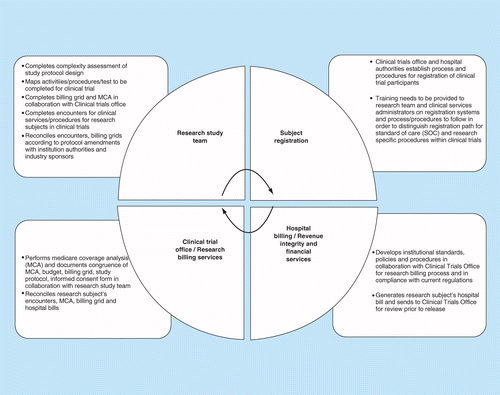
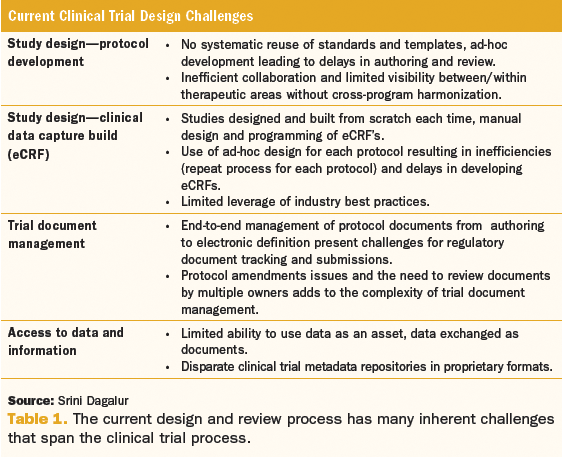
Optimization of protocol design: a path to efficient, lower cost clinical trial execution | Future Science OA

PPT – Clinical trial protocol writing: Challenges and Guidelines PowerPoint presentation | free to download - id: 906c6c-ODNhN