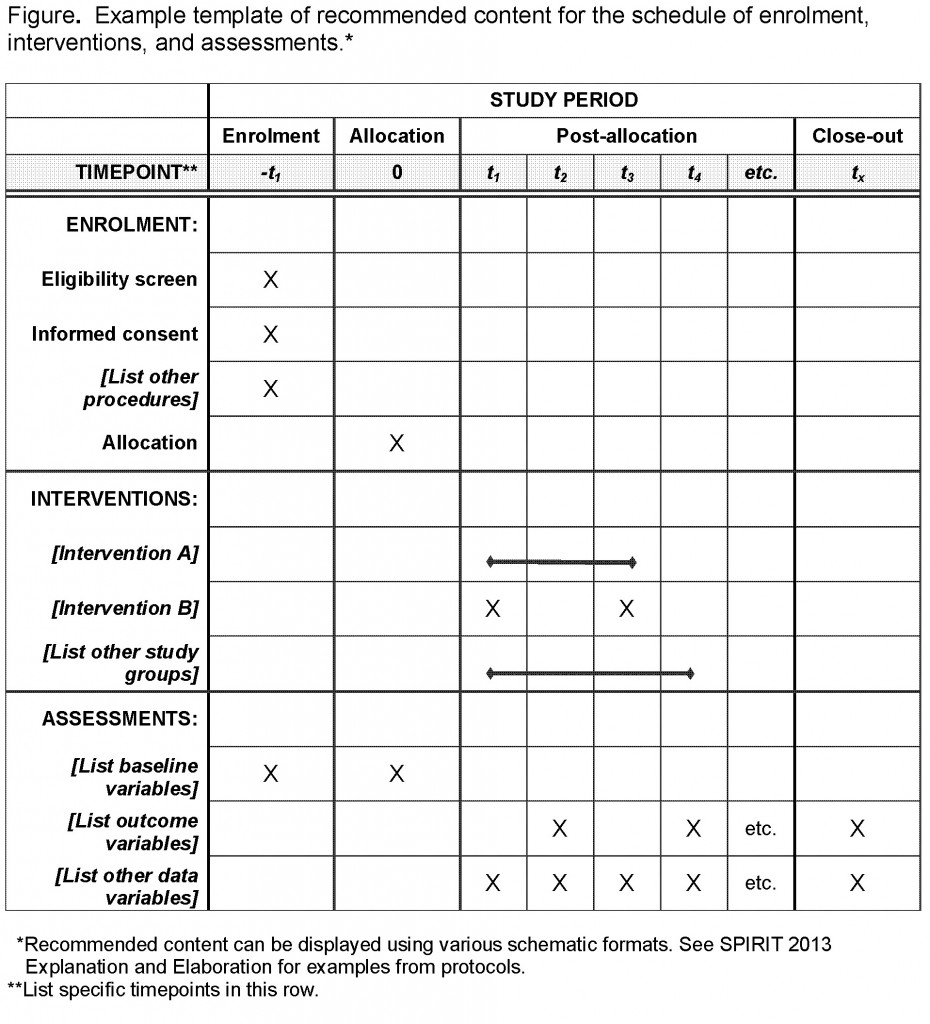
TranspariMED on Twitter: "THREAD: Today we publish a report comparing: (a) what WHO says funders should do (b) what clinical trials funders actually do Let's start with the 11 safeguards against research
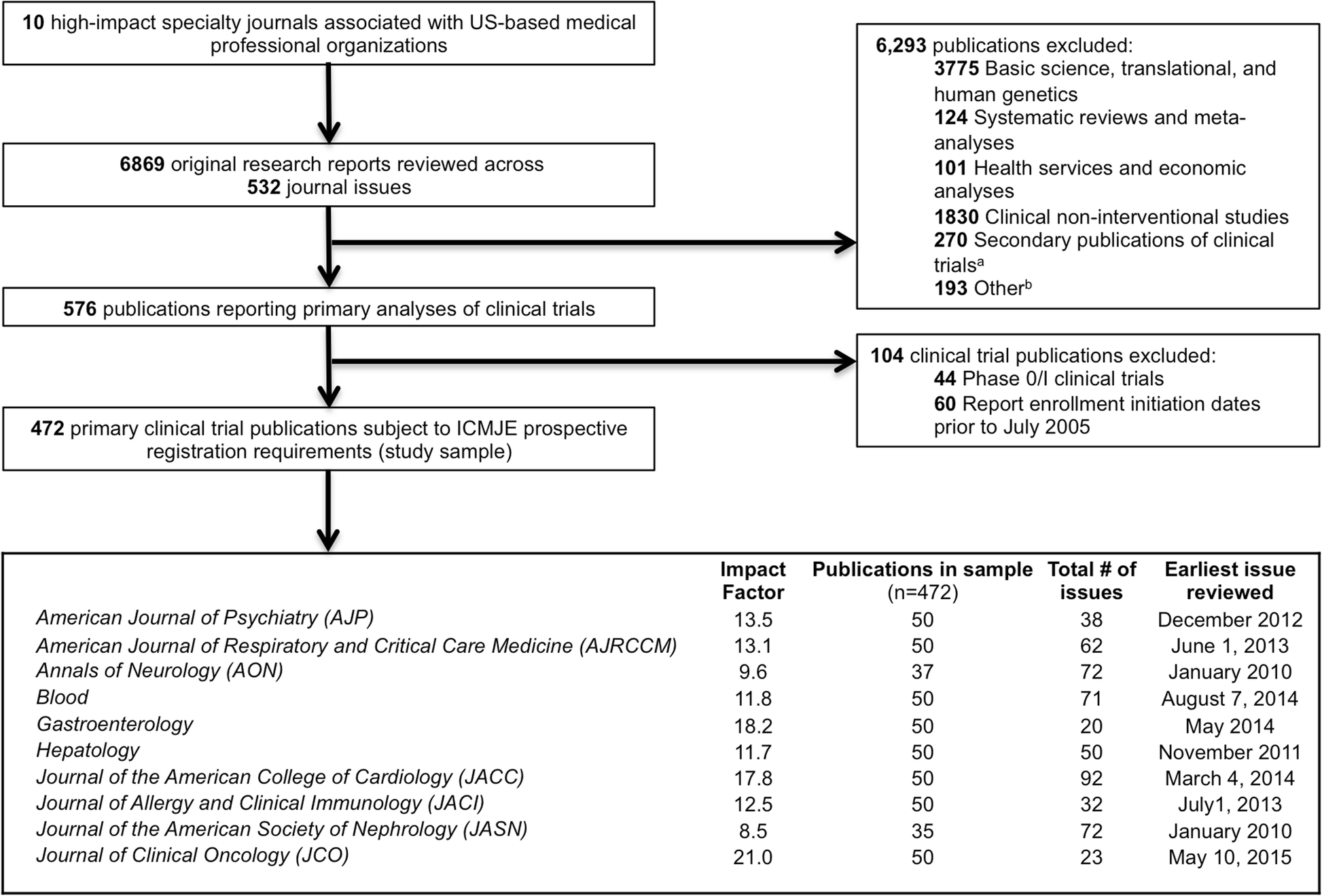
Adherence to the International Committee of Medical Journal Editors' (ICMJE) prospective registration policy and implications for outcome integrity: a cross-sectional analysis of trials published in high-impact specialty society journals | Trials

Race and ethnicity representation in clinical trials: findings from a literature review of Phase I oncology trials | Future Oncology
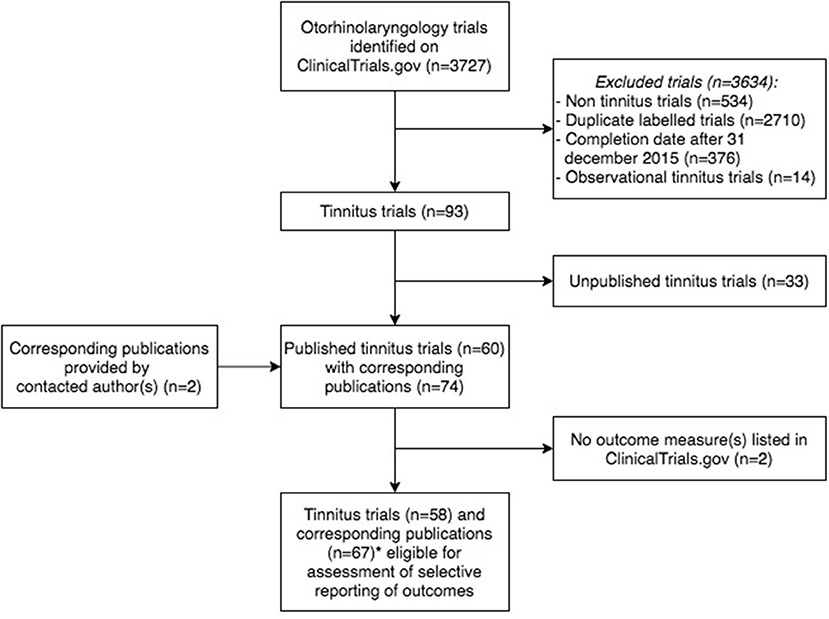
Frontiers | Selective Reporting of Outcomes in Tinnitus Trials: Comparison of Trial Registries With Corresponding Publications
![PDF] Comparison of Eligibility Criteria Between Protocols, Registries, and Publications of Cancer Clinical Trials. | Semantic Scholar PDF] Comparison of Eligibility Criteria Between Protocols, Registries, and Publications of Cancer Clinical Trials. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/62bcc7885dac29517026f3ba577045d3b93dbb7b/2-Table2-1.png)
PDF] Comparison of Eligibility Criteria Between Protocols, Registries, and Publications of Cancer Clinical Trials. | Semantic Scholar
Improving Representation in Clinical Trials and Research: Building Research Equity for Women and Underrepresented Groups | The National Academies Press

Publication and reporting of clinical trial results: cross sectional analysis across academic medical centers | The BMJ

Publication and reporting of clinical trial results: cross sectional analysis across academic medical centers | The BMJ

Comparison of serious adverse events posted at ClinicalTrials.gov and published in corresponding journal articles – The Publication Plan for everyone interested in medical writing, the development of medical publications, and publication planning