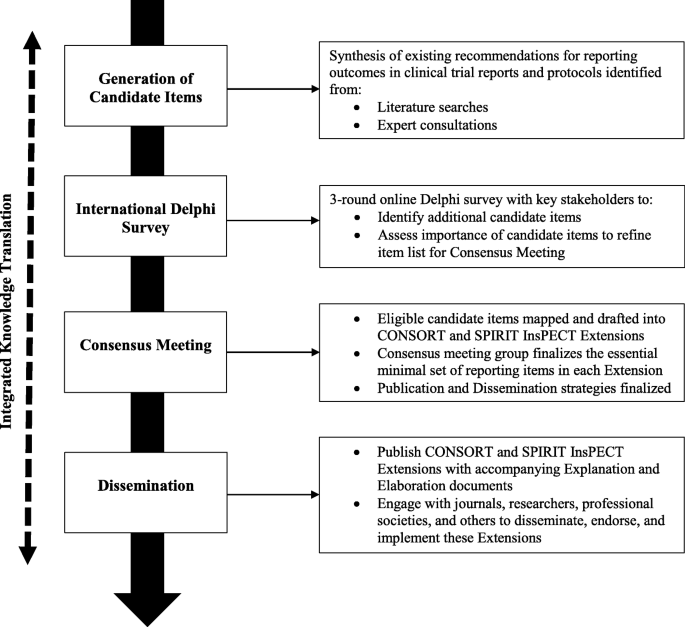
Improving outcome reporting in clinical trial reports and protocols: study protocol for the Instrument for reporting Planned Endpoints in Clinical Trials (InsPECT) | Trials | Full Text

Research2note on Twitter: "The what, when and why of research essential documents @NHSRDForum @dollyblue3 @DigitalCRN @DarkNatter @Research_Innov @WeNurses https://t.co/yPlmOQEHc6" / Twitter